Prediction of fluid viscosity of alkanes by reverse non-equilibrium molecular dynamics simulation
Theme Overview
From the perspective of curbing global warming, there is a growing demand for more energy efficient machinery and equipment such as automobiles and home appliances.
Since one way to save energy is to reduce friction loss of sliding parts, lowering viscosity is strongly necessary for fluids such as lubrication oil as well as lowering friction.
In the design of fluids, it is generally recognized that the method of molecular simulation is effective in predicting the viscosity of mixtures of more than two materials or novel fluids.
As an example, we introduce the result of viscosity calculations for common organic molecules of alkanes using PFP.
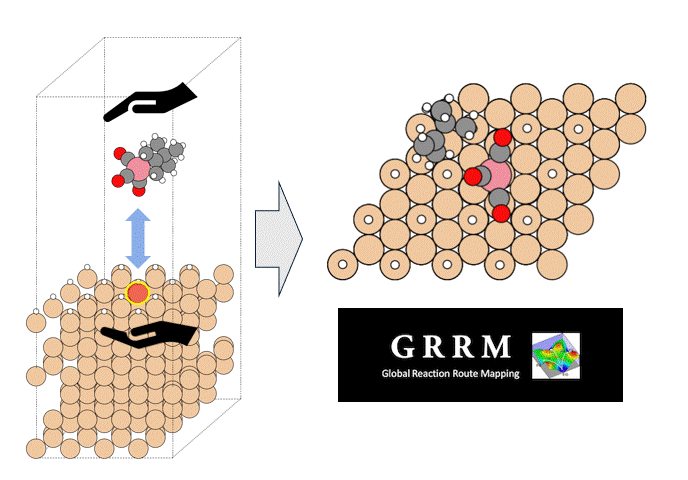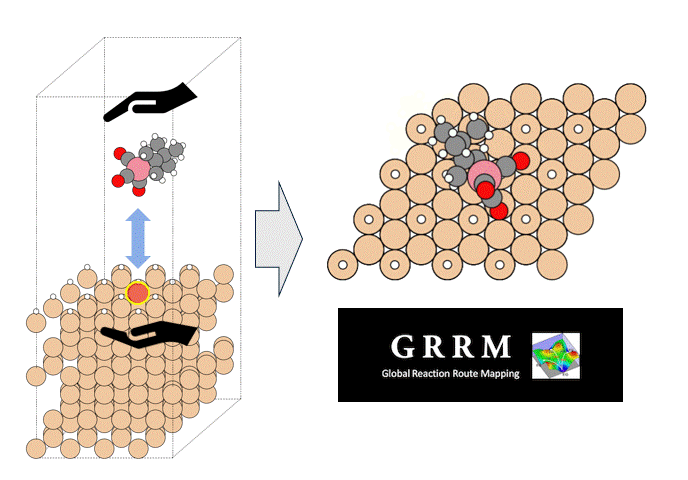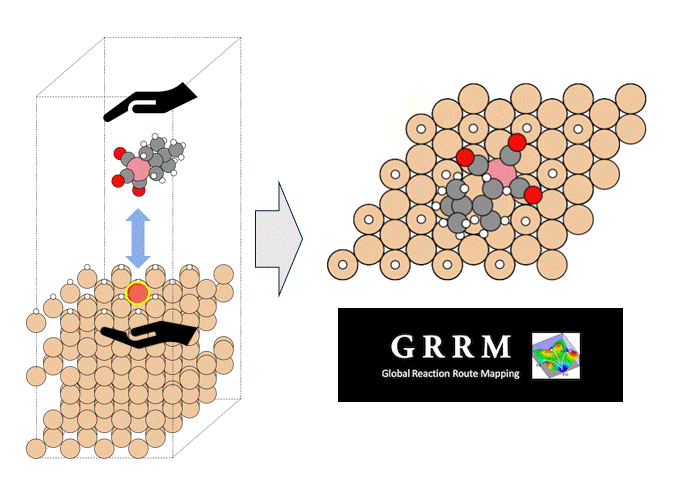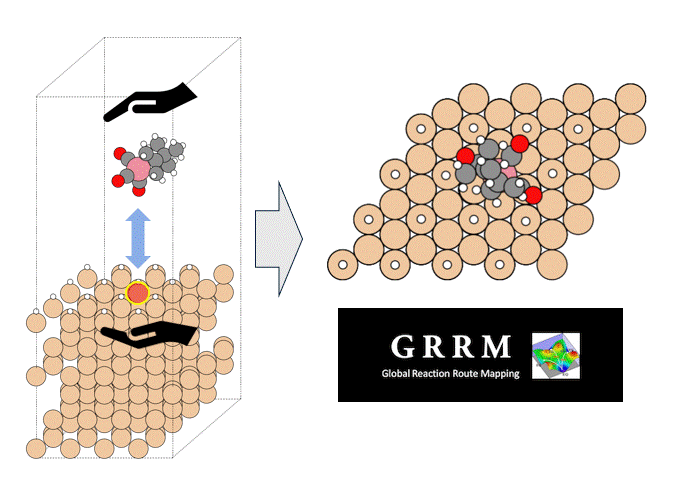
Calculation Models and Methods
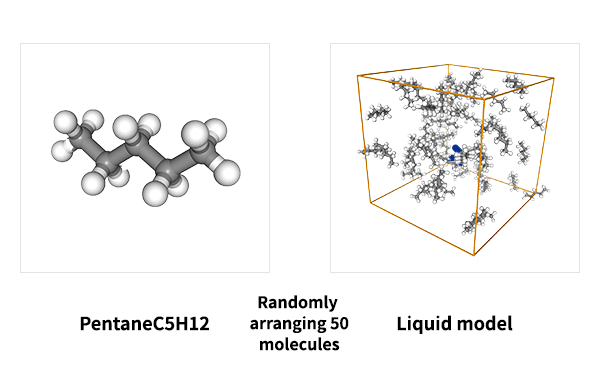
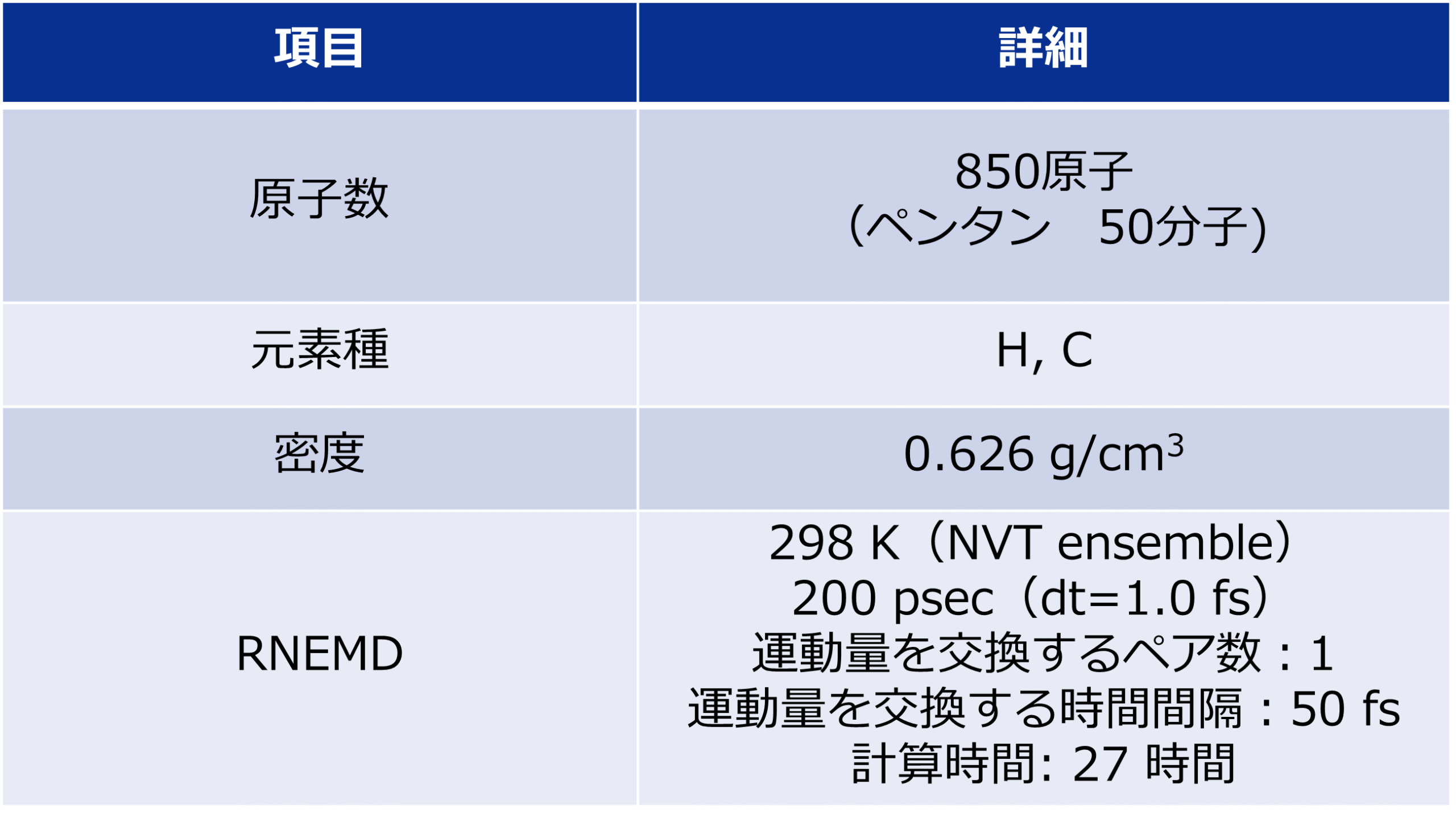
As an example of viscosity calculation, pentane, a 5C normal alkane, was selected as a target. Prior to the calculation, a liquid structure of pentane was constructed.
50 molecules of pentane were randomly placed in a simulation cell, and the cell volume was adjusted to maintain the density constant by molecular dynamics MD) method with PFP.
Three simulation models with different initial configurations were prepared to check the reproductivity of the final result.
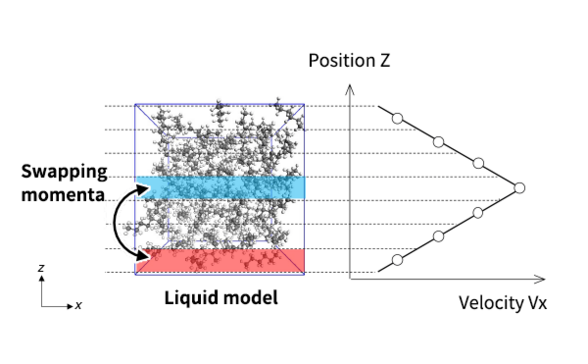
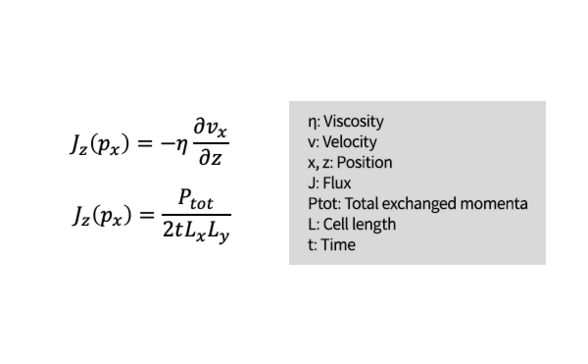
The reverse non-equilibrium molecular dynamics (RNEMD) method [1] was adapted for the purpose of calculating viscosity. This method forcibly induces velocity gradient in a liquid phase by exchanging momenta between atoms that have two opposite velocity signs, positive and negative, in two specific regions.
The RNEMD simulations were performed for duration of 2×105 steps for each of the three prepared models with different initial configurations. The integration time and temperature were set to 1 fs and 298 K, respectively.
Results and Discussion
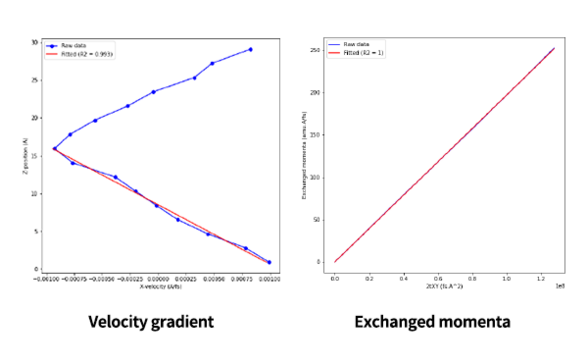

The time-averaged velocity gradient and the integrated momentum exchange rate are shown as RNEMD results for a liquid model of pentane. The velocity gradient changes linearly, indicating that the system is well in equilibrium, as does the momentum exchange.
The results of the viscosity calculation and the experimental value [2] are shown in the table. It can be seen that all three calculated values are almost the same as the experimental value. Quantitatively, the deviation from the experimental value was predicted with an accuracy of up to 14%.
This calculation was done for a simple organic compound, but our PFP possesses remarkable features of high accuracy and universality.
PFP is expected to be applied to physical property prediction of systems where a classical force field is not fixed or needs to be tuned such as ionic liquids and electrolytes.
Computational Details
References
[1] F. Müller-Plathe, Phys. Rev. E, 59, 4894 (1999). [2] J. R. Rumble, CRC Handbook of Chemistry and Physics, CRC Press/Taylor u0026 Francis.tag
Published: July 1, 2021